Puff Bar Flavors Resurface Following FDA Ban
FDA says it is aware of the situation but declined to comment on the regulatory status of disposable e-cigarette brand.
Critics say there are still gaps in regulation of marketing of vaping products.
|

The FDA is reigning in some aspects of the marketing of e-cigarettes in its finalized rule issued today, which bans their sale to minors, requires health warnings in ads and on product packages, prohibits the distribution of free-samples and requires manufacturers to show that the products meet applicable public health standards before receiving authorization from the FDA.
“The actions being taken today will help the FDA prevent misleading claims by tobacco product manufacturers, evaluate the ingredients of tobacco products and how they are made, as well as communicate their potential risks,” the agency said in a press release.
In the absence of regulation, the rapidly expanding $3.5 billion e-cigarette industry has been heavily marketing the products with questionable claims — both online and in the shops themselves — that they are safe, healthier than tobacco cigarettes and can help smokers quit tobacco, TINA.org investigations of the advertising revealed.
While a recent British Royal College of Physicians report found that the benefits of e-cigarettes, which are battery powered devices that heat flavored nicotine-laced liquids into vapor — far outweigh the potential harms and urged smokers to switch to vaping, the U.S. Preventive Services Task Force when evaluating e-cigarette studies concluded that “the current evidence is insufficient to recommend electronic nicotine delivery systems for tobacco cessation.” And several other key reports found that e-cigarettes contain harmful chemicals and can compromise immune systems in the lungs. A 2014 report by the World Health Organization, for example, stated that for some brands of e-cigarettes, the level of cancer-causing agents in the aerosol “such as formaldehyde and other toxicants like acrolein have been found to be as high as in the smoke produced by some cigarettes.”
“Today’s announcement is an important step in the fight for a tobacco-free generation – it will help us catch up with changes in the marketplace, put into place rules that protect our kids and give adults information they need to make informed decisions,” said U.S. Health and Human Services Secretary Sylvia Burwell.
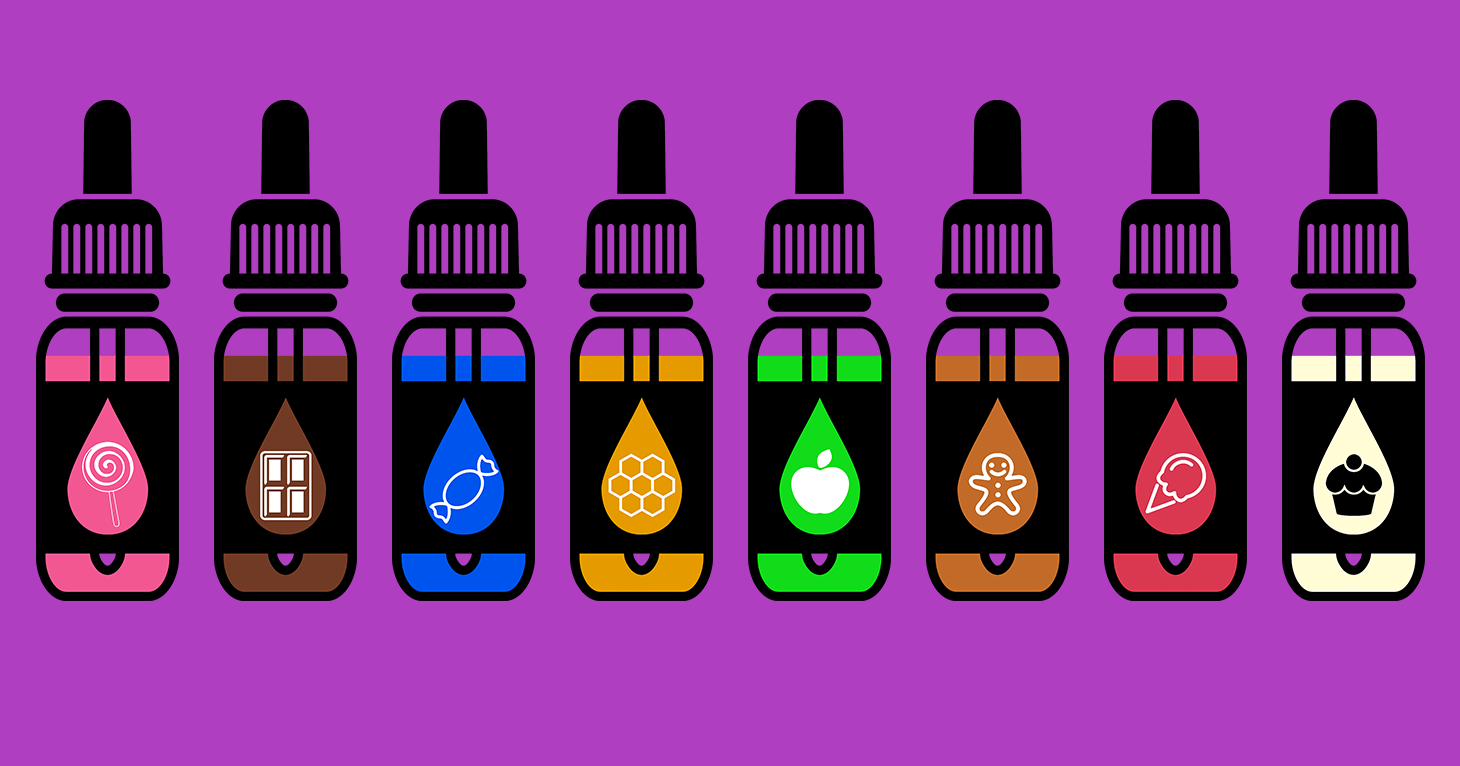
The FDA has been heavily criticized as moving too slowly to regulate the industry at a time when use of e-cigarettes, which are marketed in child-friendly flavors such as bubble gum and fruit loops, has skyrocketed. A survey by the FDA and the Centers for Disease Control and Prevention found that current e-cigarette use among high school students increased by 900 percent from 1.5 percent in 2011 to 16 percent in 2015. Advocates for regulations are concerned that youngsters could become addicted to the nicotine used in e-liquids. Studies have shown nicotine can negatively affect adolescent brains.

The finalized rule, however, does not yet restrict the marketing or flavors, nor does it take strict steps to prohibit online sales to minors, said Matthew Myers, president of the Campaign for Tobacco Free Kids, which has scrutinized the industry’s marketing to minors.
“The rule announced today falls short in protecting kids from e-cigarettes. It does nothing to restrict the irresponsible marketing of e-cigarettes or the use of sweet e-cigarette flavors such as gummy bear and cotton candy, despite the FDA’s own data showing that flavors play a major role in the skyrocketing youth use of e-cigarettes,” said Myers.
Under the rules, manufacturers of e-cigarettes and that were on the market after Feb. 15, 2007 must register with the FDA, provide details of ingredients and health risks, and are subject to inspections. It also prohibits vending machine sales if consumers under 18 have access to them. But e-cigarettes can continue to be on the market for three years while their manufacturers submit and the FDA reviews their new tobacco applications. The ban on sales to consumers 18 and under will take effect in 90 days.
The final rule, which also affects cigars and pipe tobacco, comes five years after FDA first announced it would regulate all tobacco products and more than two years after it issued a proposed rule. The rule was issued under the 2009 Family Smoking Prevention and Tobacco Control Act, which gave the FDA immediate regulatory authority over cigarettes, cigarette tobacco, smokeless tobacco and roll-your-own tobacco and also authorized it to extend its jurisdiction to all other new tobacco products.
In November, the FDA said it was opening a probe into smoking cessation claims made by e-cigarette marketers following a TINA.org investigation. The announcement came after five health advocacy organizations urged the agency to take prompt action based on TINA.org’s findings.
Lawmakers have also been urging the FTC to review e-cigarette marketing to teens and in October, the agency said it was waiting for approval from the White House Office of Management and Budget to launch such a study. Shortly after the FDA issued its final rules, U.S. Senator Barbara Boxer, D-California, urged the FTC to expedite it.
In a letter dated May 13 to the agency, Boxer said: “I commend you for studying the marketing of electronic cigarettes. But given the danger these products pose to our nation’s youth and the rapid rate at which advertising and use is growing, I also urge you to immediately act to stop these marketing tactics.”
She asked the FTC to seek out information on all forms of e-cigarette marketing including online advertising, social media, and viral videos.
In June, the FDA announced a new tool that consumers can use to report problems with vape products.
“There is no known safe tobacco product, but FDA can play a role in helping prevent certain unexpected health consequences,” said Ii-Lun Chen, M.D., director of the Division of Individual Health Science in the Office of Science at FDA’s Center for Tobacco Products in an FDA statement. “FDA wants to prevent certain unexpected health consequences that could occur from defective tobacco products, as well as health or safety problems beyond those typically associated with tobacco product use.”
Check back for updates on the FDA’s finalized rule. For more of TINA.org’s coverage of e-cigarette marketing, click here.
This story was updated on 6/9/16.
FDA says it is aware of the situation but declined to comment on the regulatory status of disposable e-cigarette brand.
TINA.org agrees with health groups that Juul’s current campaign disseminates an illegal smoking cessation claim.
Agencies crack down on copycat packaging targeting minors in the e-cigarette industry.